Case Report
Scrotal Elephantiasis Due to Lymphatic Filariasis: A Case Report Highlighting Surgical Management in a Non-Endemic Setting
- Ward Basalat
Corresponding author: Ward Basalat, Rafidia Governmental Hospital, Palestine.
Volume: 3
Issue: 2
Article Information
Article Type : Case Report
Citation : Ward Basalat, Mohammad Ahmed Abbasi, Nafe' Abu Alwan, Ola Jawabreh, Abu Saaluk Ibrahim, Laith B. Nassar, Abed Al Hafez Saleh Hassouna, Majed F.I. Alzeer Alhousseini, Ali Hanttash Abu Hanttash, Diya Asad, Majd Issa. Scrotal Elephantiasis Due to Lymphatic Filariasis: A Case Report Highlighting Surgical Management in a Non-Endemic Setting. JMCCR 3(2).
Copyright: © 2026 Ward Basalat. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.61615/JMCCR/2026/APRIL027140402
Publication History
Received Date
14 Mar ,2026
Accepted Date
25 Mar ,2026
Published Date
02 Apr ,2026
Abstract
Background
The infection, though chronic, by the nematode parasites and vector mosquitoes can cause severe lymphatic dysfunction and lead to chronic conditions such as scrotal elephantiasis. This case report elaborates on the clinical course, diagnosis, and surgical treatment of a 24-year-old male patient suffering from scrotal elephantiasis.
Case Presentation
The patient, whose past medical history includes hidradenitis suppurativa, was admitted for scrotal discomfort and swelling dating back to October 2022. The pathology was initially non-specific for reactive inflammation. A subsequent TB test was a false positive. A CT scan revealed diffuse scrotal edema with no hernia or abscess. The patient was taken for elective surgical resection of the affected tissue of the scrotum and reconstruction; he is recovering well and will have further follow-up in the clinic.
Discussion
Scrotal elephantiasis is a rare condition outside endemic areas and requires appropriate clinical and surgical treatment. The mainstay of treatment was excision with reconstructive surgery to obtain functional restoration and to reiterate the quality of life. Hence, this case has exemplified that even in a rarely presenting lymphatic filariasis, detailed diagnostic workup and purposeful surgical management form the cornerstones of treatment.
Conclusion
This often requires a multidisciplinary approach to the effective management of scrotal elephantiasis, which includes an accurate diagnosis and proper preoperative assessment with surgical intervention. This case also highlights the possibility of the occurrence of this condition in non-endemic areas and thus brings, once again, the conclusion to the forefront of the differential diagnosis.
►Scrotal Elephantiasis Due to Lymphatic Filariasis: A Case Report Highlighting Surgical Management in a Non-Endemic Setting
Ward Basalat1*, Mohammad Ahmed Abbasi2, Nafe' Abu Alwan3, Ola Jawabreh3, Abu Saaluk Ibrahim4, Laith B. Nassar1, Abed Al Hafez Saleh Hassouna1, Majed F.I. Alzeer Alhousseini1, Ali Hanttash Abu Hanttash1, Diya Asad5, Majd Issa6
1Rafidia Governmental Hospital, Palestine.
2Zagazig University, Egypt.
3An Najah National University, Palestine.
4Ovidius University of Constanța, Romania.
5Faculty of Medicine, Al-Quds University, Jerusalem, Palestine.
6Tula State University.
Introduction
Lymphatic filariasis is caused by chronic infection with nematode parasites, which are transmitted by mosquitoes. Chronic infection can lead to swelling of the extremities, hydroceles, and external genitals. It can also lead to the hardening and thickening of the skin like an elephant; it is also known as Elephantiasis [1-2-3].
The pathogenesis and clinical progression of filarial disease are affected by some factors, such as the extent and duration of exposure to infective insect bites, the quantity of accumulating worm antigen in the lymphatic vessels, the immunity of the host, and the number of secondary bacterial and fungal infections [4-5-6].
Due to the application of a global program of mass drug administration (MDA) to eliminate transmission, the epidemiology of lymphatic filariasis has decreased. MDA changed the classification of some countries as nonendemic, but more than 25 countries have been able to stop MDA due to interruption of transmission, and some appear to have eliminated transmission entirely [7].
Lymphatic filariasis can be asymptomatic or symptomatic, which appear as acute and/or chronic clinical manifestations, including adenolymphangitis, filarial fever, hydrocele, chronic lymphatic disease, and tropical pulmonary eosinophilia. Most infected residents of endemic areas are asymptomatic [8-9]
Case Presentation
This 24-year-old male had a history of hidradenitis suppurativa and presented with scrotal elephantiasis as a complication of his chronic condition. He started to develop symptoms of swelling and discomfort in the scrotum in October 2022, which were typical of scrotal elephantiasis. The patient was counselled about the need for surgical resection and reconstruction and the possibility of recurrence (Figure 1).
Figure 1: View of Scrotal Elephantiasis
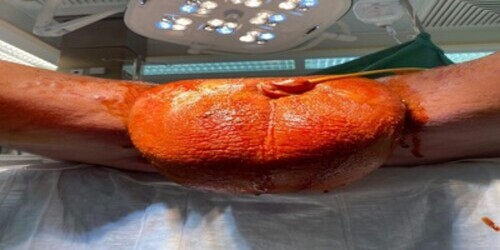
A skin biopsy was taken on 25/01/2023, and the pathology report showed non-specific reactive inflammation consistent with resolving hidradenitis; there was no evidence of malignancy or specific infectious agents.
On 05/03/2023, the patient was subjected to a TB test as part of the preoperative assessment; this result showed false positives, probably due to the low likelihood of infection with M. tuberculosis given the clinical presentation and lack of exposure history.
A CT scan of the abdomen and pelvis was done on 25/10/2023 to define the full extent of scrotal involvement. Also, it has excluded other possible complications: steady diffuse edema of the scrotum; no evidence of hernia or abscess formation is noted. All abdominal organs and lymph nodes are unremarkable.
He was admitted on 24/10/2023 for elective surgical resection of the affected scrotal tissue with subsequent reconstruction (Figure 2). Treatment was aimed at being symptomatic and improving the quality of life. Intraoperatively, a split-thickness skin graft from the left thigh was used for reconstruction. This type of graft typically takes 10–12 days to confirm no rejection, and it has a good vascular bed.
During surgery, a circular incision was made at the base of the penis to achieve a good dissection and elongation of the penis. The key aspect of the operation involved resecting all lymphatic tissue from the scrotum to prevent any recurrence of the problem. A new pouch was created in place of the scrotum using the healthy skin from the dilated scrotum, ensuring no lymphatic disorder.
Figure 2: Post-Operation View of the Scrotum
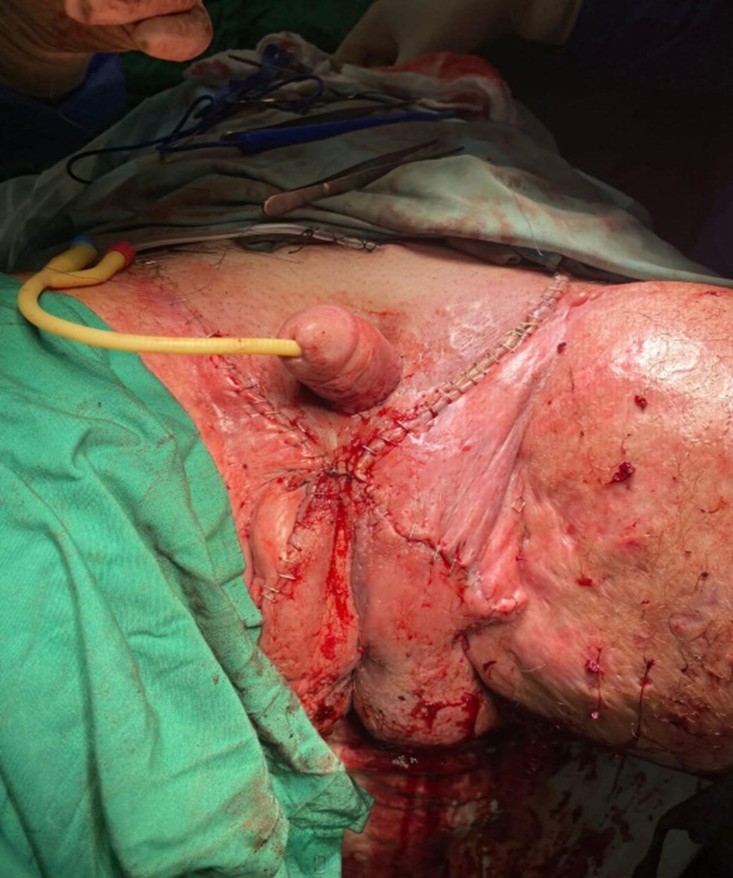
In the postoperative period, he was monitored for complications and reviewed regularly about his wound and antibiotics. He spent 9 days in the hospital. During this period, daily wound dressing and assessments were done. The patient improved decently with absolutely no signs of infection or other complications. At discharge, he was placed on oral antibiotics with clear instructions on wound care. Follow-up with the urology department was also arranged after his discharge to check on the healing process of the wounds. The patient now has a normal sexual and psychological life that has changed for the better.
Discussion
Massive scrotal lymphedema, also termed elephantiasis, can be caused by obstruction, aplasia, or hypoplasia of lymphatic vessels [10]. It is rare even in areas endemic to filariasis [11]. It is usually caused by acquired infection, e.g., lymphogranuloma venereum or filarial infestation with Wuchereria bancrofti. Scrotal elephantiasis is scarce outside endemic regions in Africa and India. Occasionally, it has been attributed to radiotherapy, neoplasm, and lymphadenectomy [10,12,13,14].
Primary lymphedema, i.e., congenital elephantiasis, is expressed at different ages, and it may be sporadic or inherited. Inherited diseases constitute approximately 15% of the cases of congenital lymphedema. Patients develop edema in adolescence without restriction to the external genitalia. In hereditary elephantiasis of the Meige type, lymphedema of the external genitalia occurs due to malformation of lymphatic vessels. In some cases, hidradenitis suppurativa and lichen sclerosis et atrophicus have also been described [15,16].
Etiologic factors that lead to lymphedema of the external genitalia are infections, neoplasms, or chronic inflammation. It is rarely caused by congenital conditions, e.g., the rare Meige syndrome leading to malformation of lymphatic vessels of the external genitalia [13].
In lymphatic filariasis, the main lymphatic channels become dilated, and flow is impaired. The network of peripheral lymphatics in the skin becomes a ‘safety valve’ and acts as a conduit through the skin until healthy lymphatic trunks are found proximally to the damaged vessels. Lymph stasis and lymphotention lead to the accumulation of interstitial fluid, protein growth factors, and other active peptide moieties, glycosaminoglycans, and particulate matter, including bacteria. Consequently, there is an increase in collagen production by fibroblasts, an accumulation of inflammatory cells (predominantly macrophages and lymphocytes), and activation of keratinocytes. This process results in protein-rich edema fluid, increased deposition of ground substance, subdermal fibrosis, and dermal thickening and proliferation.
Patients mostly present with painless swelling. Gradually, the skin becomes coarse and tough as fibrosis sets in. This massive enlargement of the genital skin can cause various symptoms, including disfigurement, urinary dribbling, impotence, and recurrent cellulitis, along with emotional disturbance.
It may lead to scrotal lymphedema, causing irreversible damage to the involved tissue and posing a risk of necrotizing fasciitis.
Treatment consists of restoration of lymphatic drainage by lymphangioplasty. It has been attempted with limited success in minor cases without fibrosis. In more advanced cases, the mainstay of treatment consists of excision of all the affected skin and soft tissue. The procedure is sometimes called reduction scrotoplasty. The posterior skin of the scrotum can be used as a skin graft on the base of the penis.
Possible operative complications include hemorrhage, injury to the urethra, hematoma, wound infection, and recurrence of lymphoedema.
Conclusion
This case represents a patient with scrotal elephantiasis due to lymphatic filariasis, its diagnostic challenges, and strategies of effective management. Detailed pre-operative assessment by pathology, imaging, and clinical evaluation was done to plan a course of treatment, including surgical resection with reconstruction. This marked a tremendous improvement in the quality of life for the patient; on average, no postoperative complications or recurrence were noted during the follow-up period. The case thus highlights the need for a multi-disciplinary approach in the management of such rare and severe manifestations of lymphatic filariasis for the best possible patient outcomes. Additionally, documenting such cases is crucial, especially in non-endemic areas, so that, through improving awareness, diagnostic and therapeutic strategies can be changed for this rare condition when outside typical endemic regions.
- Rebollo MP, Bockarie MJ. (2017). Can Lymphatic Filariasis Be Eliminated by 2020? Trends Parasitol. 33(2): 83-92.
- Maldjian C, Khanna V, Tandon B, Then M, Yassin M, Adam R, Klein MJ. (2014). Lymphatic filariasis disseminating to the upper extremity. Case Rep Radiol. 2014: 985680.
- Shukla SK, Kusum A, Sharma S, Kandari D. (2019). Filariasis presenting as a solitary testicular mass. Trop Parasitol. 9(2): 124-126.
- Baird JB, Charles JL, Streit TG, Roberts JM, Addiss DG, Lammie PJ. (2002). Reactivity to bacterial, fungal, and parasite antigens in patients with lymphedema and elephantiasis. Am J Trop Med Hyg. 66(2): 163-169.
- King CL. (2001). Transmission intensity and human immune responses to lymphatic filariasis. Parasite Immunol. 23(7): 363-371.
- Lamb TJ, Le Goff L, Kurniawan A, Guiliano DB, Fenn K, Blaxter ML, Read AF, Allen JE. (2004). Most of the response elicited against Wolbachia surface protein in filarial nematode infection is due to the infective larval stage. J Infect Dis. 189(1): 120-127.
- World Health Organization. (2021). Global programme to eliminate lymphatic filariasis: progress report.
- Freedman DO, de Almeida Filho PJ, Besh S, Maia e Silva MC, Braga C, Maciel A. (1994). Lymphoscintigraphic analysis of lymphatic abnormalities in symptomatic and asymptomatic human filariasis. J Infect Dis. 170(4): 927-933.
- Nutman TB, Kazura J. (2011). Lymphatic Filariasis. In: Tropical Infectious Diseases: Principles, Pathogens and Practice, 3rd ed, Guerrant R, Walker DH, Weller PF (Eds), Saunders Elsevier, Philadelphia. 729.
- Nelson RA, Alberts GL, King LE. (2003). Penile and scrotal elephantiasis caused by indolent Chlamydia trachomatis infection. Urology. 61: 224.
- McDougal, W.S. (2003). Lymphedema of the external genitalia. (Review). 170(3): 711-716.
- Kuepper D. (2005). Giant scrotal elephantiasis. Urology. 65(2): 389.
- Mc Dougal WS. (2003). Lymphedema of the external genitalia. J Urol. 170(3): 711–716.
- ammer ME, Plogmeier K, Schneider W. (2002). Surgical therapy of scrotal edema in elephantiasis congenita hereditaria (Meige type) Urol A. 41(5): 493–495.
- Smeltzer, D. M, Stickler, G. B. and Schirger, A. (1985). Primary lymphedema in children and adolescents: a follow-up study and review. Pediatrics. 76(2): 206-218.
- Kinmonth, J. B, Taylor, G. W, Tracy, G. D, Marsh, J. D. (1957). Primary lymphoedema. Clinical and lymphangiographic studies of a series of 107 patients in which the lower limbs were affected. Br J Surg. 45(189): 1-9.
Download Provisional PDF Here
PDF




p (1).png)




.png)




.png)
